Notes from the Long Covid Definition symposium at the National Academies of Sciences, Engineering, and Medicine.
Last week I was honored to deliver the keynote address to kick off the second day of the Symposium on Long COVID: Examining the Working Definition, hosted by the National Academies of Sciences, Engineering, and Medicine (NASEM) in Washington, DC.
The event featured an impressive diversity of voices, from patients and advocates, to laboratory scientists, clinicians, to big data epidemiologists. As well it should have. The event was part of NASEM’s ongoing effort to examine and help refine the definition of Long Covid—at the request of the Department of Health and Human Services, no less. No small task.
My lecture was entitled “The therapeutic validation of Long Covid,” and it was based on my recent publications in the Lancet Infectious Diseases and which you may have already read about here in Inside Medicine.
But in preparing my talk, I began to notice something. It seemed to me that those tasked with the challenge of improving the definition of Long Covid would soon have to confront a brewing central tension that could stand in the way of progress. Should the definition of Long Covid be more permissive and therefore include more people? Or, should the definition be more restrictive?
Pros and cons of “permissive" diagnoses.
The parameters of disease definitions are not mere technicalities. They have profound implications. Take diabetes. Let’s imagine we decide that starting tomorrow a patient can be diagnosed with diabetes if their HemoglobinA1C level (the main blood test we use) is 5.9% or higher, instead of 6.5% which is the current cutoff in most places. Doing this would have two immediate effects. First, in an instant, millions of people would suddenly have diabetes. Second, the disease soon would look less deadly because we would have added millions of people to that community, but none of whom who were sick enough to die from it. If you keep adding more people with low “A1c” levels, eventually the new and expanded diabetes population would not have any more risk than the general population—and what would be the point of that?
When you look at Long Covid, it’s not so simple. We don’t have a simple blood test or scan so far. It’s all about symptoms and timing. There are multiple competing definitions. If one definition, say, requires more than 6 months of symptoms, it likely excludes a number of people who have Long Covid. But if a definition requires just 3 weeks of symptoms, it likely includes a number of people who probably don’t have Long Covid.
The growing tension.
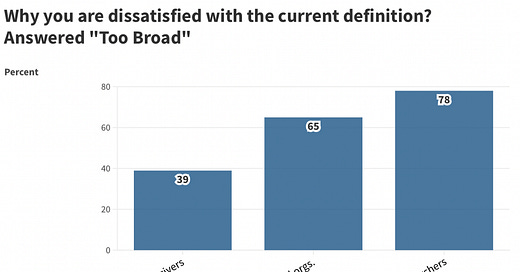
Take a look at results from a poll whose results were distributed to the attendees of the NASEM Long Covid symposium.
Patient and patient organizations (advocacy groups, etc), are generally not worried that the current definitions of Long Covid are too broad (i.e. permissive). Only 39% of these respondents think that’s a problem. But researchers are more concerned about the definition being too board. Twice as many researchers as patients responded that the current Long Covid definition is too broad.
Why? Probably because the researchers believe that the more inclusive the definition is, the more heterogeneous the patients who enroll in studies will be. That will make the disease more difficult to study, and the search for effective treatments more elusive. Imagine trying to study a chemotherapy regimen that probably works against lymphoma but the population of volunteers in the study is dominated by patients with other forms of cancer in whom the drugs simply won’t work.
The graph above that I created from my slide deck makes this point visually. The broader and more inclusive a disease definition is, the less likely scientists will be able to find a treatment, precisely because separating the signal from the noise in a well-conducted trial becomes increasingly insurmountable.
But there’s an equally worrisome possibility that I fear may also be forgotten in all of this. Another way to demonstrate the tradeoff is to invert it—looking at the likelihood that scientists might mistakenly conclude a drug does not work for a condition it actually treats effectively. The more inclusive the definition of a disease is, the more likely that would be to happen—again because a statistical signal for a therapy that works would get drowned out.
The way forward.
As you can tell, in preparing this talk, I became increasingly convinced that my role as something of an outsider—neither a Covid-19 patient, advocate, nor a full-time Long Covid researcher—was to bring attention to this potential conflict over whether the Long Covid definition should be broader or narrower. The goal of this was not to drive the parties apart, but rather to acknowledge this tension and encourage everyone at the table to resolve the issue collaboratively.
The analogy I ended up using was this: Do we want one big tent or lots of little ones? One big tent would mean a broad and inclusive definition. Lots of little tents would mean multiple narrower definitions for the different subtypes of Long Covid. (Indeed, Long Covid is unlikely to be “one solitary entity.”)
One big tent would feel inclusive. But it could also get impersonal with individuals getting lost in the crowd. Meanwhile, life in little tents may feel a bit lonelier at times, but would ultimately be more personal. Again, if I developed, say, lymphoma, I’d want to be seen by lymphoma doctors (and meet in patient support groups), with people going through the same thing. It would be a disservice for me to be seen by a doctor who specializes in lung cancer, and get together with patients going through something entirely different than me.
So, I did put my thumb on the scale in the direction of moving towards multiple definitions of Long Covid that reflect the various subtypes that I expect researchers will identify.
Therefore, my parting message to the work group tasked with refining Long Covid definitions was to 1) wish them success on the important task that lies ahead of them and; 2) to invoke our national motto: E pluribus unum. Out of many, one.
When the video of my talk is available, I’ll share it. In the meantime, I welcome your comments and feedback.






Our health system uses Epic electronic medical record. ICD 10 codes already exist and I have been using them during outpatient visits for long Covid syndromes. Normally there is a long Covid umbrella code, but also more specific subsets like Long Covid with neurological manifestations. Lately I’ve been using “Post Covid condition.” Too many options, but a standard nomenclature that allows broad coding when symptoms are broad, and more narrow symptom defined nomenclature when symptom cluster in one body system would be best IMO. And what about PASC?
Alternatively, we could ask Starbucks for permission to use their drink sizes: Short, Tall, Grande, Venti Hot, Venti Cold, and Trenta. This may have a disarming and empowering effect with an otherwise fearful condition.
I’m in a little tent, an orphan of sorts. My symptoms ( which I didn’t have before Covid) are not mentioned anywhere. 14 months later still have sinus pain , sometimes headache or teeth ache, and on my worse days a general feeling of unwellness in addition to my sinus sx. Sinus CT was totally normal which was a huge blow
Today is a good day. Yesterday bad because I wore a mask on a long plane trip . Last day of no symptoms since the start was March7. I am hesitant in saying I have LC because others are far worse off than I. So I understand the dilemma