“What about Long Covid?” This is the most common question I get whenever I discuss the fact that Paxlovid has become far less useful for most people than it was back when it was first released.
Last week, a much-anticipated Long Covid study was released in JAMA Internal Medicine. Researchers recruited volunteers with Long Covid and randomized them to receive either Paxlovid or placebo for 15 days. The goal was to find out whether Paxlovid reduced or eliminated Long Covid symptoms 10 weeks later.
Bottom line: There was no effect.
Patients’ symptoms improved similarly in both the Paxlovid and the placebo group upon entry into the study. The study was stopped early because the statisticians declared it futile based on pre-planned “interim” analysis.
Who was studied, what was found and some (potentially alarming) findings.
As with any data, the results apply to the particular population that was studied. Here are some features:
Average age: 43.
Average time with Long Covid: 17 months.
Body types: a majority of patients were in the overweight or obesity categories.
Severity of Long Covid symptoms: moderate to severe.
Percent of participants who were vaccinated prior to the study: >98%
Percent of participants who took Paxlovid or another antiviral during their acute illness: 26%.
Percent of participants hospitalized during their acute illness: <6%.
The primary outcome of the study was overall improvement across six Long Covid symptoms (fatigue, brain fog, shortness of breath, body aches, GI symptoms, and cardiovascular symptoms).
But this particular list of six symptoms was somewhat arbitrary (albeit, pre-determined). So, I was particularly interested in whether Paxlovid might have helped any of the individual symptoms, rather than the sextet of symptoms as a whole.
Answer: Nope.
In fact, in the study’s appendix, I noticed that the odds that Paxlovid recipients had fatigue or brain fog increased by the end of the study (which was actually 15 weeks, though the primary outcome was symptoms after 10 weeks). While this is technically a secondary finding, and therefore can only be used to formulate a hypothesis for a future study, rather than as a firm conclusion, if it were to be confirmed in another study, it would be a devastating blow to the notion of using this drug for treating Long Covid. The authors didn’t mention this in the actual prose of the paper, though they did acknowledge that the percent of patients whose brain fog improved (to mild or none) was lower in the Paxlovid recipients. I found this a bit alarming. Remember, for reasons we do not always understand, some drugs we hope will work can do nothing or cause harm. This suggests the possibility of that, but is hardly definitive on that point.
Long Covid cause implications.
If this study had been successful, it could have had important implications for the search for the cause of Long Covid. In particular, Paxlovid is an antiviral medication. If the drug had precipitously improved Long Covid symptoms, it would have pointed strongly towards hypotheses related to either chronic live virus or viral persistence (in which remnants of the virus, including its genetic material, might be causing symptoms). That said, the study’s failure to show a benefit does not rule anything out. It just means this particular approach didn’t work.
That said, the researchers did try to identify signs of chronic infection in this cohort of patients by way of stool testing. None of the patients’ samples were positive for SARS-CoV-2 on PCR tests.
The devil is in the definitions.
If Long Covid is actually several distinct entities (which seems likely), it’s possible that some drug could help people with one subtype of the disease and not another. In fact, that was the crux of my talk to the National Academies of Sciences Long Covid Symposium a year ago, which gathered a body of experts to generate a new definition of Long Covid.
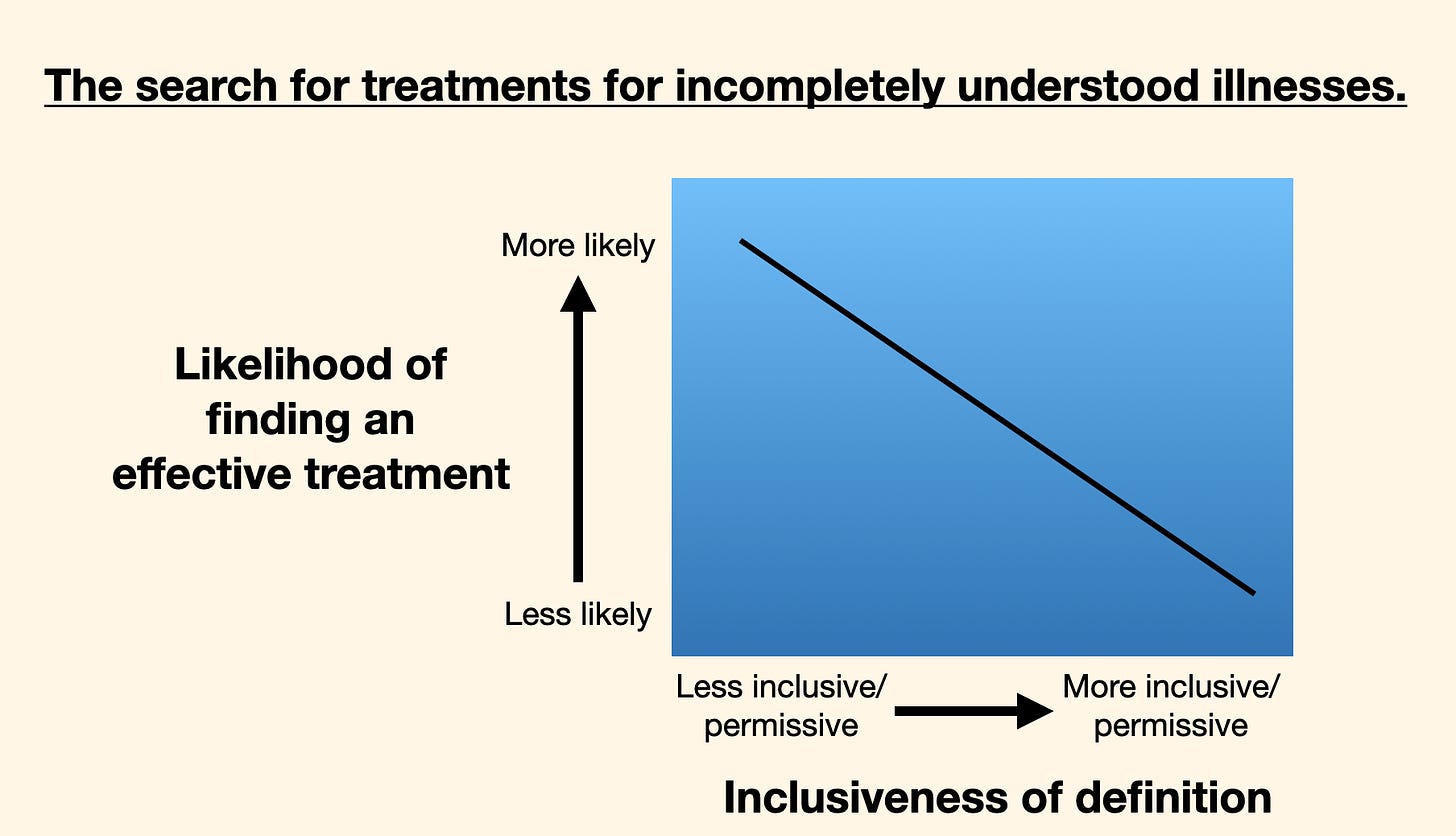
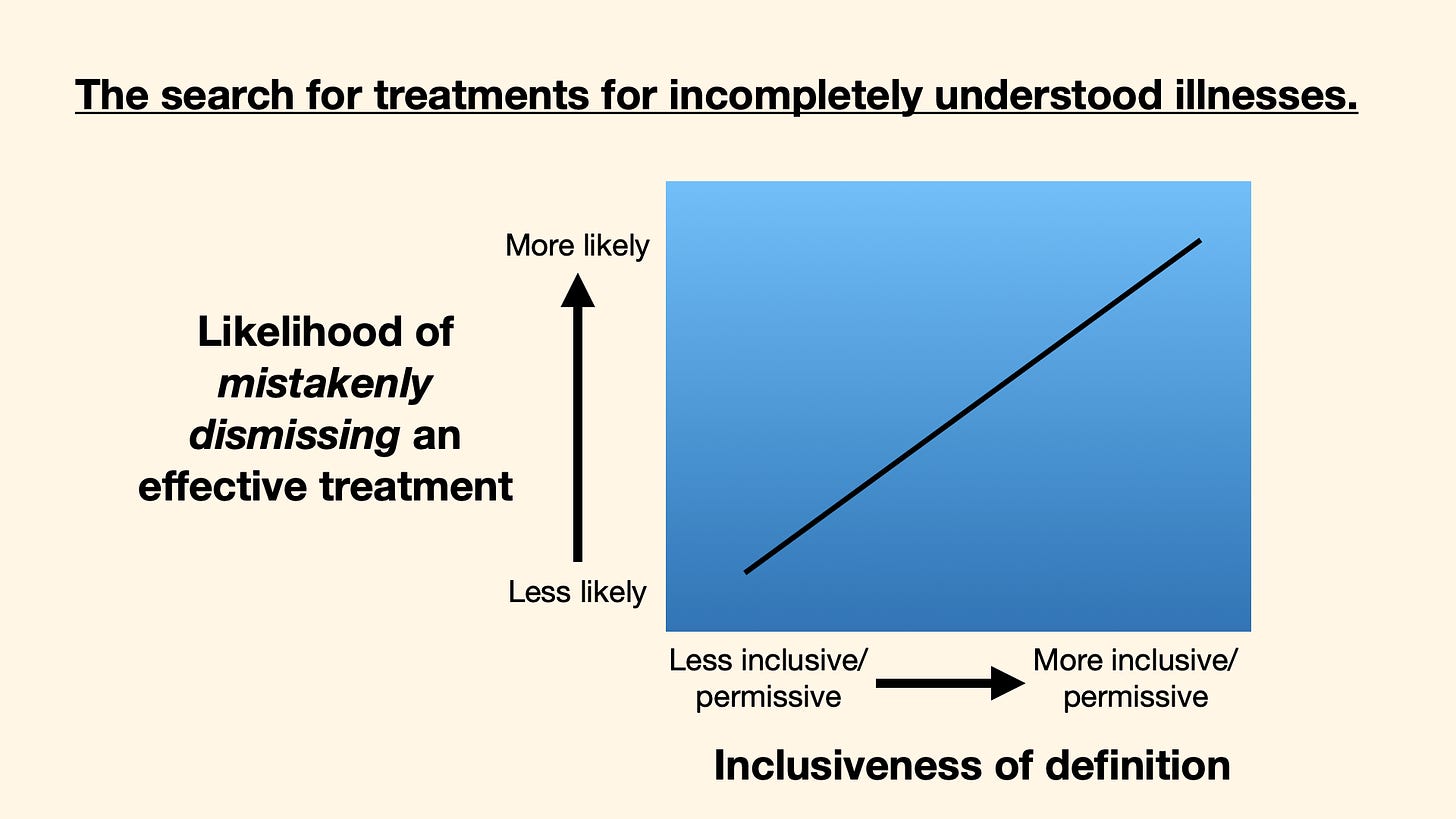
My request to that august body was that they balance the need to be inclusive (“more inclusive/permissive”) with the need to be specific (“less inclusive/permissive”). The idea was analogous to if we tried to treat all cancers with the same regimen, we’d almost certainly always fail. But if we tested a particular regimen for a specific cancer, we could succeed.
Below are two slides from my talk. They show a problem around disease definitions. If a definition is too inclusive (big tent), researchers might have trouble finding a treatment that works for everyone who meets the broad definition. And just as importantly, a big-tent definition increases the chances that researchers might mistakenly dismiss a treatment that actually works for some of the patients.
What’s next?
There is another major study of Paxlovid as a possible treatment for Long Covid underway (led by my mentor and friend Dr. Harlan Krumholz and his Yale colleague Dr. Akiko Iwasaki). That study is decentralized (i.e., not at one hospital, like the one above), and will also attempt to measure immune system markers of disease that may be present and modifiable.
Am I optimistic that it will show a benefit for Long Covid? Not particularly.
Do I think we can learn other important things from it and other studies that makes the effort worthwhile? Absolutely.




Given your recent experience with Covid & this new study (and other recent studies), under what circumstances would you ever recommend Paxlovid to a fully updated vaccinated person of any age?
Dr. Faust, Good thoughtful and informative essay. It is evident of a lot of careful thinking and work. Thank you.