CDC updates RSV vaccine guidelines for older adults, leaves infant policies unchanged.
Your "Five on Friday" lead story for July 5, 2024...
We are back with “Five on Friday", the feature where Inside Medicine behaves like an actual medical/health/science newsletter. Also, please vote in the poll at the end!
If you value this service—and Inside Medicine in general…
Here we go…
Item 1: CDC updates its RSV vaccine guidelines. Here’s the bottom line.
For decades, there was no vaccine for RSV (Respiratory Syncytial Virus). Now that we have them, the question is who needs doses and how often? The answer comes down to identifying those at risk of serious outcomes and how long vaccine protection lasts. Scientists are still sorting that out, so I expect the CDC’s recommendations to change over the next few years as we learn more. This week, the CDC updated its guidance based on the latest data it has. Starting now, here’s who the CDC says should receive an RSV vaccine:
All people ages 75 and older.
People ages 60–74 with increased risk of a severe RSV-related outcome.
Now, just what “increased risk” means remains vague, but it includes patients who “have certain chronic medical conditions, such as lung or heart disease, or [who] live in nursing homes.”
Please note that the CDC is not saying that people in these groups need an annual RSV vaccine. So, if you already got one last year, you do not need another one—at least that’s the current CDC stance.
The reason for this is that we do not yet know how long vaccine-derived protection lasts for important outcomes. The implication is that the favorable effects of RSV vaccination last longer than those of Covid or flu shots (which the CDC recommends people receive each year), which could reflect differences in how long our bodies’ immunity lasts and differences in mutation rates across these viruses (albeit, also which outcomes epidemiologists are tracking in their decision-making.)
As always, these decisions reflect risks and benefits. There have been concerns about side effects, particularly a rare but important one called Guillain-Barré Syndrome. For older adults recommended to get RSV vaccines, however, the benefits have thus far outweighed the risks. (That said, any future annual shot policies would have to be analyzed for how much benefit and how much risk gets added by such an approach.)
Meanwhile, the guidance for infants (the other group the CDC has made RSV recommendations for) remains unchanged this year:
Infants under 8 months old (i.e., those entering their first RSV season) should receive nirsevimab (an antibody infusion) if their mother did not receive an RSV vaccination during pregnancy.
Children ages 8–19 months at increased risk of severe RSV disease should receive nirsevimab.
Item 2: Supreme Court upends Chevron framework. Healthcare implications have already begun to unfold.
Last week, the Supreme Court overturned a policy (known as Chevron) that had deferred to the expertise of government agencies when a federal law needed interpretation. It took less than a day for the healthcare sector to pounce. On June 28, a New Jersey-based healthcare system sued the Secretary of the Department of Health and Human Services, stating that he had incorrectly interpreted the meaning of some Medicare policies. The plaintiffs argument is that HHS’s interpretation of things has deprived their hospital of reimbursements.
This is the tip of the iceberg, I would think. There are an enormous number of actions that HHS takes in regulating hospitals. With the Chevron framework now gone, it may be open season for lawsuits against the federal government. The effects of this change is hard to predict. But already, a wide range of possible implications are being discussed by experts (this initial story by Shannon Firth for MedPage Today gives an early taste), from prior FDA drug approvals to rules that aim to keep nursing homes safe. A great deal of uncertainty (if not chaos) around all of this is to be expected in the coming months and years.
Item 3: Standing orders increased emergency contraception.
With abortion access under attack in various ways in the US in the post-Roe era, some places have taken steps to push back. Massachusetts is one such place. Within a couple of months of the Dobbs decision, Massachusetts enacted “standing orders” for emergency contraceptive pills like “Plan B” and “Ella”. That means that a person asking a pharmacist for one of these drugs does not need a specific prescription to obtain the pills. A new study shows that the policy worked as intended. After the policy was enacted in August 2022, fills of emergency contraceptive went up 32%. In particular, the Ella option went up primarily—which makes some sense because the window of effectiveness for Ella (5 days) is longer than that of Plan B (3 days). Such a policy would be even more important in places with new abortion bans that begin after just a few weeks of pregnancy. And for women who do not want to be pregnant, taking emergency contraception (instead of a medical or procedural abortion many weeks later) is a lot less burdensome.
Item 4: A new Covid-19 antiviral? Maybe.
Obviously it would be good to have another potential tool for treating Covid. We might be a step closer. Recently, Pfizer shared results of its phase 2 trial of a new Covid-19 antiviral, ibuzatrelvir at a medical conference. The question is whether this drug works and whether it will accomplish what we need it to accomplish.
So far, the drug “works” in one sense—recipients had lower SARS-CoV-2 viral loads than those who received placebo. What we don’t yet know is what that means. Paxlovid also lowers viral loads, but that has not led to improvement in outcomes (including severity or duration of symptoms) in standard-risk patients. Meanwhile, at least so far, Pfizer has only shared data on ibuzatrelvir for adults <65 years. Notably, people with immune compromise were not included in the study. So, I’m wondering what gap this drug is supposed to fill. Is it hoped that it will do a better job at controlling symptoms? Is it supposed to reduce hospitalizations in younger adults who already have very low rates of it? One upside of the drug is that it is hoped that it would have fewer drug-drug interactions than Paxlovid. But that begs the question; why is Pfizer studying this drug in non-geriatric adults (i.e., those not likely to be taking drugs that might have unsafe interactions with Paxlovid)? I’ll update you on this when we have more data, but I’m a little concerned that Pfizer is studying the wrong population here. While it would be nice to have an antiviral that works for younger people (perhaps decreasing missed days of work, or doing what Paxlovid has not been able to and decrease symptoms), our larger concern should be for the highest risk populations. I don’t know what Pfizer’s phase 3 trial looks like, but I hope they’ll be studying the populations who need a Covid treatment the most.
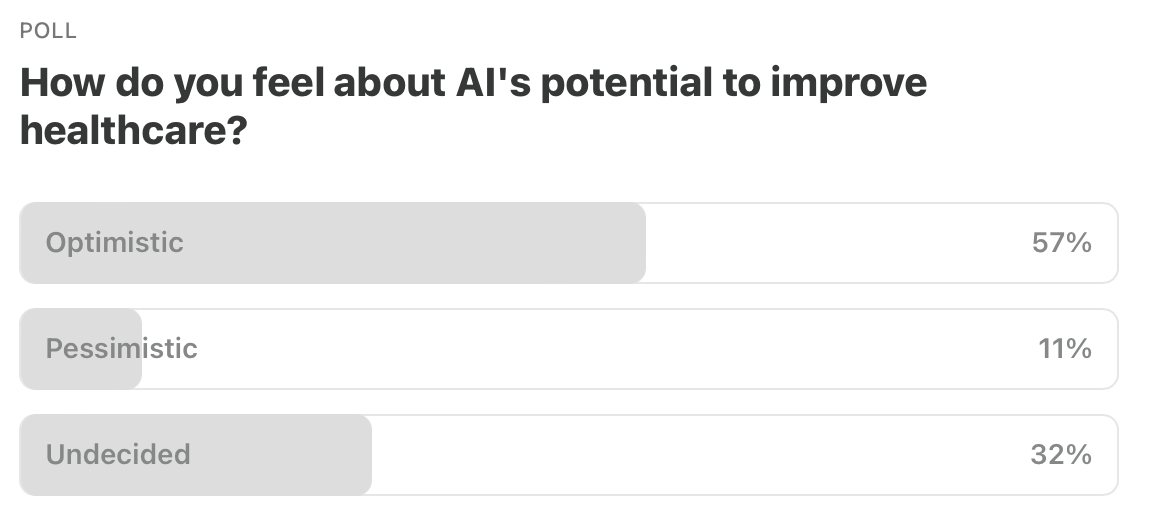
Item 5: Poll of the Week.
Here are the results from last week’s poll. Thanks for your votes—and your nuanced comments.
Item 5a: Poll of the Week for this week!
Okay, I’ll admit that I’ve seen almost none of last week’s Presidential debate. I’ll probably watch it to see for myself. That said, I trust your judgment, Inside Medicine community. Leaving aside the complete unfitness for the presidency that his opponent has (sorry if you disagree, but y’all know how I feel about our previous President)…
That’s it. Your “Friday Five!”
Feedback! Do you like the “Five on Friday” format? Have any ideas for next week’s Poll of the Week? Any great articles you read elsewhere that you want to share with the Inside Medicine community? Other musings or thoughts?
Please contribute to the Comments!





Not concerned about his ability to do the job - he’s got a very competent team in place to help execute policy. VERY concerned about his electability now - I don’t like switching horses this late, but think we need to do whatever we can to field the strongest possible candidate. And he no longer is perceived as “ok to go” by enough voters, so that means he should step aside.
I'm sorry to say that I find the poll is really badly designed. A single evening's event is no basis whatsoever for a determination of someone's fitness for officer, and posing it this way just makes the question seem like a valid one - when it is irrelevant except for the media's focus on it. It is ridiculous to think that the process of choosing a replacement would lead to anything other than the election chaos the repugs would delight in, and that would very likely lead to a further four years of chaos. (Though as you point out, the Chevron decision alone will create a great deal of uncertainty, not only in health care, and thus create a fair amount of chaos for a long time to come - until there's a different composition of the court, probably.)