Ozempic and Drug Holidays: Taboo or the Next Frontier?
Taking breaks from taking obesity drugs might work--despite how experts have interpreted the data--and that might help with costs and shortages. Research is needed.
The blockbuster obesity drugs— Ozempic, Wegovy, Mounjaro, and others—are changing lives. Along with causing substantial weight loss, we’ve seen a parade of studies showing that these medications reduce deaths, strokes, heart attacks, heart failure, and kidney failure (the data on the last one were so strong when the data were analyzed midway through the study, that the trial had to be stopped early, because it was deemed unethical to withhold the drug from the placebo recipients). And as I’ve written, I suspect the downstream effects of these medications will continue to surprise us in good ways. I predict that knee and hip replacements will become less common, to name just two possibilities.
But from the first time I heard about these medications from obesity medicine experts, something bothered me: their absolute unwillingness to discuss the notion that patients may be able to stop taking these medications at some point, or use them intermittently.
The rationale for this appears to be two-fold:
“Obesity is a disease.” The thought here is that patients shouldn’t think of Ozempic and the others as quick fixes (i.e., diet pills).
Most studies show that most patients do indeed start to gain weight back when they stop taking these drugs.
The problem with the current dogma on obesity medications.
Specialists want patients and the public to remember that obesity is a chronic disease, not just some social problem. I get that, and I agree with it. But it does not necessarily follow that patients must therefore be consigned to a lifetime of taking these medications (which do have side effects, and are extremely costly). Look, patients with high blood pressure, diabetes, or high cholesterol are absolutely told to stop taking their long-term medications if and when their numbers are consistently low enough without the help of the pills to warrant that.
So I’m seeing some misdirected advocacy here. Yes, obesity is a disease. But that does not mean that treatments must be for life for all patients.
Now, the better rationale for keeping patients on these medications in the long-term are the data showing that weight gain predictably occurs after cessation.
But that analysis is inadequate and incomplete. I want to talk through the possibility that “drug holidays” or dose modifications should be studied in the future, and discuss what that might mean.
Drug holidays. A taboo which must be studied.
Drug holidays are planned breaks from taking medications. Drug holidays can be used to assess the ongoing need for treatment. For example, if a patient taking blood pressure pills has a safe blood pressure even when they don’t take them, why continue them? Drug holidays can also decrease resistance/tolerance to the medications, meaning they’ll work for longer. Drug holidays also decrease costs.
Let’s use a figure from a recent article in the Journal of the American Medical Association that studied tirzepatide, another GLP-1 agonist like Ozempic and Wegovy.
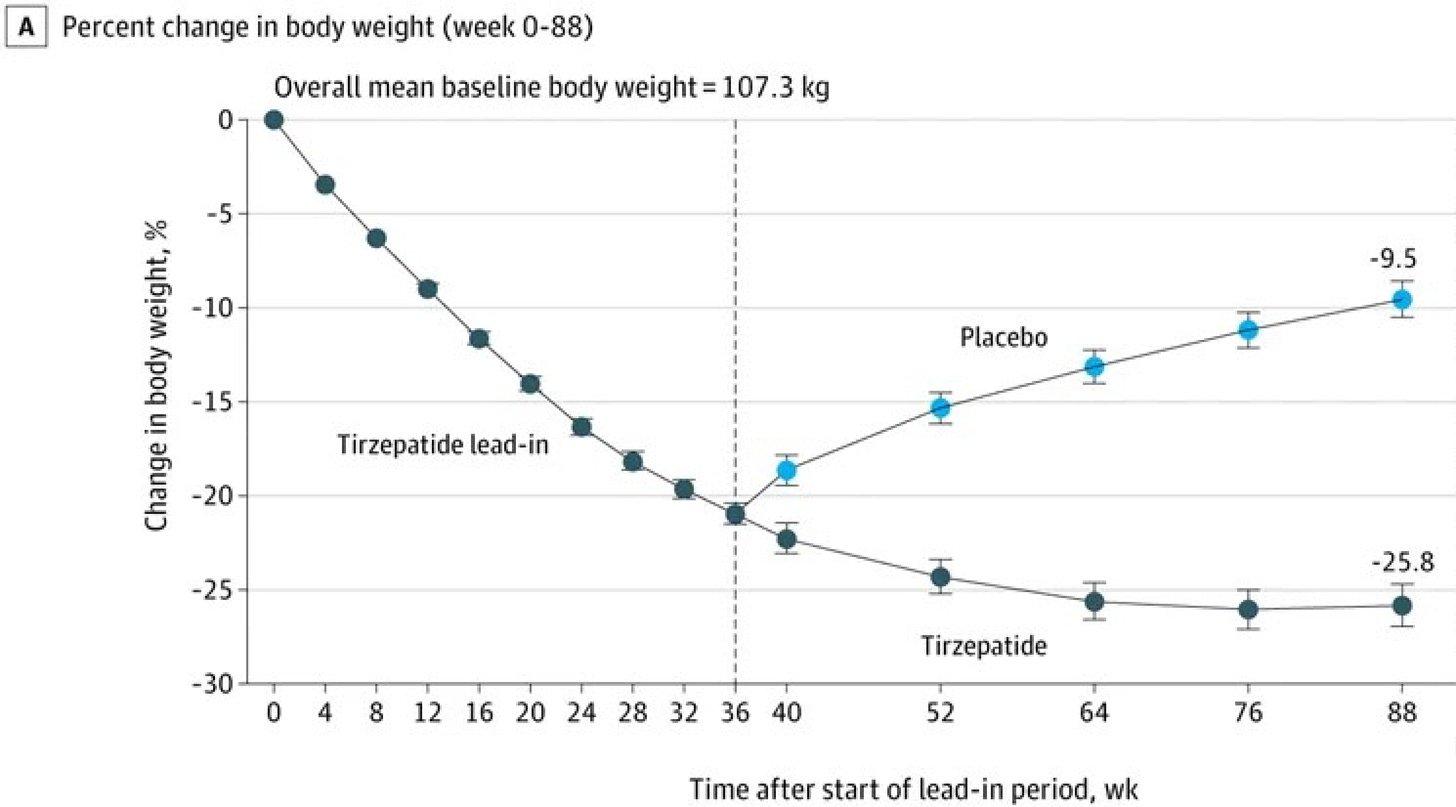
First, let me explain it. Then, I’ll annotate it and dig further. The above graph shows percent change in body weight (Y-axis) over the 88 weeks of the tirzepatide study (X-axis). In this study, all participants received tirzapatide for 36 weeks. Then, half kept taking it and half took placebo, for another 52 weeks. It’s obvious that everyone lost weight in the first 36 weeks—and quite a bit; a 21% decrease, from 236 lbs on average, to 188 lbs. That’s remarkable for 8.5 months. But from there, we can see that placebo recipients gained back about half of what they lost over the next year. This (and data like it) serves as the basis for the contention that once patients with obesity start taking these drugs, they must stay on them forever.
But what if that’s not true? What if most of the health benefits of that weight loss would be realized if people lost and maintained >10% of their starting weight, not the >26% loss that the tirzepatide patients had achieved by week 88.
In fact, in a recent study of Ozempic that I highlighted in Inside Medicine, death rates (and heart failure exacerbations) in patients with cardiovascular disease decreased markedly after they lost and maintained around 10% of their initial body weight for a couple of years.
What is the real point of these blockbuster obesity medications?
And this is the crux of the argument. What if the real point of these blockbuster obesity medications is to keep patients out of some “danger zone” for most of their future lives. Let’s say—for argument—that for the participants in the JAMA tirzepatide study, the cardiovascular and other metabolic risk factors (diabetes, kidney problems), drop substantially if they are able to consistently maintain a >10% decrease in body weight from their starting points, and that getting to >15% or >20% loss is not really necessary. To understand the implications, I have annotated the graph from before (red shade=more risk, corresponding to being closer to original weight and blue shade=less risk).
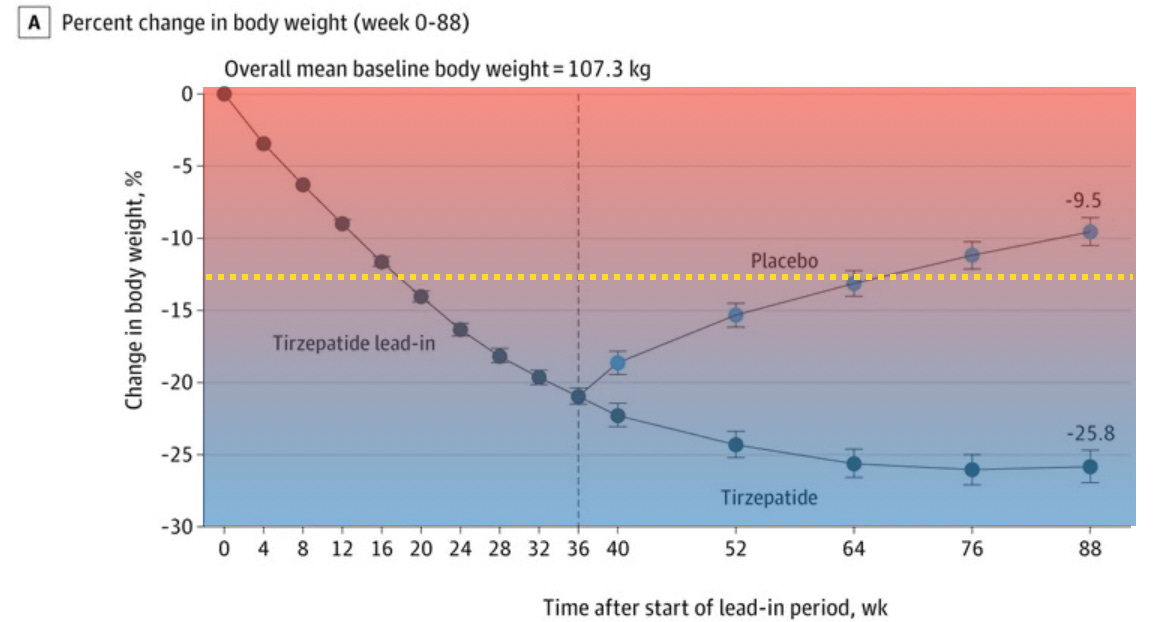
Notice, also, that I’ve added a yellow dashed line. Think of that new line as a hypothetical “Rubicon” or line in the sand. Our goal is for patients to spend most or all of their time below that yellow dashed line of badness. (I drew the yellow dashed line at -12.5% from initial body weight—the thought being that if a patient reaches that point, they’ve gotten “too close” to the -10% barrier where the cardiovascular and metabolic risks start re-emerging.)
Now let’s imagine a situation in which our goal is to keep a patient in the safe zone after taking tirzepatide for 36 weeks. To do this, we’ll allow drug holidays whenever our patient is well below the yellow dashed line, but resume the drug when they cross it. We can envision an average study participant as the use-case. Our patient starts taking tirzepatide at week 0. He loses 21% of his body weight by week 36. He then stops taking the drug, because the “induction period” has ended and he’s lost more than enough weight to be in the safe zone. By around half a year later, though, he has predictably gained back around 8% of the 21% of the original body weight he had lost. At that point, he’d have reached that yellow dashed line on the graph—too close to the -10% barrier. As his doctors, we would advise him to resume taking the drug, perhaps for another 6 months, and then see where he is. Over time, we might find that this patient is able to stay out of the danger zone 100% of the time simply by taking the drug half the time (after an initial 36-week induction period).
The benefits of this would be extremely important. First, the cost of the drug would be cut in half in all years after the initial induction. Second, the patient would be less likely to develop tolerance to the drug’s effects, making it more useful in the long-run. Third, it might give the patient a chance to see how other lifestyle modifications are working, now that he has been given a running start, so to speak.
What we do not know—and what we need to learn.
The question is, where do we draw these lines? When can we (when should we) stop and resume these medications? How long can patients live at -15% versus -25% of their initial body weight without sacrificing the cardiovascular and metabolic benefits of weight loss.
Nobody knows. The reason is that this has not been studied—in part because of a taboo, a dogma that says that these drugs “must be taken indefinitely.” I hope I have convinced you—including some experts and clinical trial designers who read this newsletter—how important it is that abandon this dogma and study drug holidays. If the concept works as I outlined here, we could treat twice the number of patients (alleviating shortages) for the same price (so, really, at half price) while achieving the same astounding health benefits we have seen in a recent series of landmark studies.
Questions? Comments? Leave them in the Comments section below…



My husband is on Ozempic for diabetes that suddenly became very poorly controlled: it's been amazing and he's had significant weight loss. His PCP and me (a family doc) have debated how to take the least amount of the medication as his numbers have normalized. There is absolutely no data on the .25 dose which the company claims doesn't work on hyperglycemia but observation says it does. An off ramp or downward taper is needed. And to make all the doses the same price is just price gouging.
Another option: try spreading out the doses. How sure are we that 7 days is the magic number? A patient could mark the calendar, take the drug every 8 days, every ten, etc. At every two weeks they’ll be spending half as much, and their weight may oscillate by 1-2% instead of 10%.