New bivalent booster results for seniors from Israel are good news for everybody. Key questions remain.
Bad outcomes are becoming more unusual. Boosters continue to help, at least in the short-run.
Booster update. Let’s review new data on the bivalent booster (the new version of the Covid-19 mRNA vaccine which contains some surface proteins resembling the original virus, and some resembling Omicron variants). Today, we’ll cover clinical data from real patients living in the world. Tomorrow, I plan to untangle a debate that is heating up about antibody levels detected in laboratory studies.
The most interesting clinical data so far are out of Israel (from a new non-peer-reviewed preprint posted on the Lancet website). Israel’s bivalent booster rollout began in September, so this is a fast turnaround.
Let’s cut to the chase:
The study shows evidence of good short-term protection against Covid-related hospitalization—to the tune of an 81% reduction, among bivalent booster recipients ages 65 and older. The outcomes only follow patients for about 70 days so far.
Just 2% of the hospitalizations occurred among patients who were boosted recently (all with the bivalent formulation) even though boosted people comprised 14% of the study populatoin. That’s great news for the booster. (Although, wow, booster rates are low in Israel, especially for in this older population).
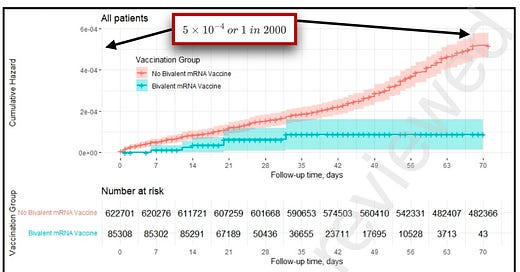
In more good news overall (and not just for the booster), this study is yet another example of a trend in which bad outcomes are becoming less common in Covid studies. Of 622,701 patients ages 65 and up followed in the Israel study, just 295 of them had a Covid-related hospitalization. That’s one Covid-19-related hospitalization for every 2,111 people studied, regardless of booster status. That’s not high for this age group, especially considering where we’ve been in the past. The rate of hospitalization in both the boosted and non-boosted group was so low that the Y-axis was literally labelled in scientific notation (see below), which is how researchers write tiny (or huge) numbers.

Data from Israel on the bivalent booster. By 70 days after receiving the bivalent booster, only around 1 in 2,000 recipients had been hospitalized for Covid— around 81% less than those not recently boosted. Annotations: Inside Medicine. Let’s put this into context. In a study out of Qatar from spring of 2022, 1 in 185 patients studied (vaccinated and boosted combined) had severe, critical, or fatal Covid-19. Now, it’s not exactly fair to compare these figures to the new Israeli data, since the Qataris follow more stringent cutoffs for what constitutes “bad outcomes.” (The Israelis record hospitalization rates, not necessarily severe cases per se; In other words, there are patients in Qatar who were hospitalized with Covid-19 who were not deemed “sick enough” to be counted as a severe case in their studies.) However, it’s undeniable that the rates of serious short-term outcomes are lower in our most recent major Covid studies (as we’ve discussed). In fact, the same authors of the new bivalent booster study found similar rates of hospitalization last winter and spring in a study of Paxlovid as the Qataris did last spring. So, it’s nice that bad outcomes are lower in this latest paper, regardless of booster status. (Although, again, the recently boosted people were far less likely to be hospitalized.)
We expected to see a decrease in hospitalization from a study like this, at least for a couple of months, anyway. The question is whether the bivalent formulation will mean longer protection than we saw with prior booster doses. That’s simply unknown for now.
We have no idea how much of the decrease in hospitalization is bound up in temporary protection against infection, but it’s probably a lot of that. You can’t be hospitalized for Covid if you don’t get Covid—and the boosters certainly decrease infections for some time. Interestingly, the Israeli researchers did not report outcomes on infection. In fact, I asked the lead author about that this evening, and he told me they have not even bothered to look at infection rates yet.
What none of this tells us is whether the bivalent booster performs better than the monovalent booster we were using before. These latest results compared bivalent booster recipients to people who had not received any booster lately. So these data cannot be used to say that the bivalent booster is better than the monovalent booster we were using before this fall. Recent data out of the CDC indicate that bivalent boosters become more effective in preventing infection the further out from your most recent dose someone is, due to waning. Again, not surprising, but also not evidence if bivalent booster superiority.
Summary:
Boosters yet again have been shown to decrease Covid-19 hospitalizations among older adults (and likely people of all ages with substantial medical conditions). We knew this about the monovalent boosters and now we know this about the bivalent ones. What these data do not tell us is whether the new bivalent boosters are better than the monovalent ones they replaced. We’ll likely never get a clinical head-to-head trial that would settle that, which is disappointing. But we’ll at least soon know if the protection lasts longer with the bivalent formulation. If the benefits of the bivalent booster fades less quickly, that would be indeed evidence of a real and clinically relevant advantage over the old recipe. I’m eager to find out.
Bad outcomes from Covid-19 seem to be becoming increasingly less common in major studies of vaccines and therapeutics overall. As bad outcomes become more unusual, we’ll have to be careful to sort out which populations need more boosting, and which ones can (or even should) eventually skip additional doses that may be coming down the pipeline.



Thank you for this. I am in for five and hoping we get to one a year for those who need it or for all of us. Really hoping this virus stops throwing curve balls. We all need a break!
Waiting to hear if seniors should receive another boost of the bivalent after 6 months…